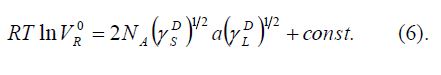courtesy of Surface Measurement Systems Ltd
- The surface energy is a useful parameter describing the energetic properties of the surface of a solid sample. It can be determined in a fast and accurate way by IGC SEA. This paper describes the measurement of the dispersive component of the surface energy of Paracetamol.
Introduction
The surface energy is an important parameter for the characterisation of surface properties. It can provide a useful picture of the energetic situation on the surface and shows therefore a strong dependency on various macroscopic properties. For instance the dispersive component of the surface energy is a useful tool to follow changes on oxide surfaces caused by temperature treatment [1] or to highlight batch-to-batch variations in pharmaceutical production [2].
An easy way to study such effects is the use of dynamic methods. IGC SEA is a particularly suitable technique that allows a fast and accurate determination of the surface energy, either the dispersive component or the interaction with a polar probe.
In the current study N-Acetyl-p-aminophenol is used as a model substance to measure the dispersive component of the surface energy by the elution of an alkane series in the infinite dilution range.
N-Acetyl-p-aminophenol (Acetaminophen, 4-Acetamidophenol) is the active component of the well-known drug “Paracetamol” (Figure 1).
Commercially available Paracetamol contains additional ingredients supporting the tableting of the active component. Therefore tablets were also measured for comparison under the same conditions.
Theory
In an IGC SEA pulse measurement an injection of the adsorbate is made. This pulse is transported by the carrier gas, which for the SMS-iGC SEA is helium, through the GC to the column. The amount adsorbed in the column is eluted by the carrier gas.
These measurements are carried out at infinite dilution where only very few probe molecules are available for the interaction with the surface.
For this reason only the highest energy sites on the surface are covered which provides the highest sensitivity of the measured parameters.
In the infinite dilution range peaks are symmetrical (Gaussian) and the retention volume can be calculated from the retention time at the
peak maximum. The net retention volumes V0R are computed using Equation 1.
where T is the column temperature, m is the sample mass, F is the exit flow rate at 1 atm and 273.15K, tR is the retention time for the adsorbing
probe and t0 is the mobile phase hold-up time (dead time). j is the James-Martin correction, which corrects the retention time for the
pressure drop in the column bed. The relation between the retention volume and free energy of sorption ΔG is given by Equation 2.
where R is the gas constant and K is a constant depending on the chosen reference state (De Boer or Kemball/Rideal) [3]. Moreover
ΔG is related to the energy of adhesion WA (between probe molecule and solid) by Equation 3 (as an approximation).
where a is the cross sectional area of the adsorbate and NA the Avogadro constant. According to Fowkes [4] WA can be split into two
terms (Equation 4):
with WAD denoting the van der Waals forces and WAS the specific, mainly polar interactions. Subsequently the retention volume is a measure
for both components. However, the measurement of polar interactions will be discussed in a later application note. In the case of dispersive interactions WA is given by Equation 5.
with γsD and γLD as the surface tension of the adsorbent and the adsorbate.
Two methods are described in literature to calculate γsD which is the dispersive component of
the solid surface energy. The method of Schultz et al [3] uses a plot of ΔG versus a (γLD)1/2 for a series
of alkanes. Combining Equations 3,4 and 5 leads to:
An alternative method is based on the value of ΔG for a series of n-alkanes [5]. This leads to the expression
where αCH2 is the surface area of a CH2 unit (6Å2) and γCH2 is its free energy (35.6 mJ/m2).
Method
Two different columns, one with a 2 mm ID and one with a 3 mm ID, were packed with N-Acetylp-aminophenol, supplied by Sigma-Aldrich (purity
99.0%). Tablets of “Paracetamol-ratiopharm” were used from Merckle, Germany, which contained 78 wt% of N-Acetyl-p-aminophenol.
The tablets were crushed and the powder was packed in a 2 mm ID column.
All the sorption experiments were carried out on an SMS-iGC 2000. Measurements of the dispersive interaction were made with 3%
undecane, decane, nonane, octane and heptane at 303 K (all solvents from Aldrich, HPLC grade). A pretreatment was made for 1 h at 303 K, 0% RH.
Results
Figure 2 shows the surface energy plot obtained from a measurement on a 3 mm column at 30°C.
The calculation of the surface energy was made according to Schultz et al [3] by the SMS-iGCanalysis software (v1.1). The obtained values for all columns are listed in Table 1.
The 3 mm column was measured twice to check the reproducibility.
Table 1. Experimental obtained dispersive surface energies.
The reproducibility in the same column is excellent (only 0.5% deviation). This also suggests only a small dependence on pretreatment time as well as an exclusively reversible interaction. The difference of 1 mJ/m2 (2.5% deviation) between the values of the 2 mm column and the 3 mm column shows a very good agreement between the different column sizes and packing. The value of the 2 mm column which contained the tablet powder gave a more significant difference to the others but is still in good coincidence.
The average value of all measurements on NAcetyl-p-aminophenol is 40.13 mJ/m2 compared to 33.94 mJ/m2 in the case of the tablets.
Obviously the tableting ingredients and processing significantly lower the surface energy.
Conclusion
Surface energies may be readily studied using an IGC at infinite dilution. In the particular case of 4-Acetamidophenol the dispersive component of the surface energy was determined with a good reproducibility. Paracetamol tablets showed a smaller surface energy due to the additional tableting ingredients and processing methods.
Acknowledgement:
SMS thanks Frank Thielmann and David Butler for their contributions to the Application note.
References
[1]Papier, E. and Balard, H., Chem. Mod. Surf. 3 (1990), 15
[2] Ticehurst, M.D., Rowe, R.C. and York, P., Intern. J. Pharm.111
(1994), 241
[3]Schultz, J., Lavielle, L. and Martin, C., J. Adhesion 23 (1987), 45
[4]Fowkes, F.M., Ind. Eng. Chem. 56 (1964), 40
[5]Dorris, G.M. and Gray, D.G., J. Coll. Interf. Sci. 77 (1980), 353